 The House-passed Build Back Better Act would have done this for all vaccines (including COVID-19 vaccines). Policymakers could ensure access to COVID-19 vaccines for all Medicaid enrollees in many ways, but one sensible approach would be to extend vaccine coverage rules similar to those that apply to the Medicaid expansion population to other Medicaid adults. For the time being, states are subject to a temporary requirement to cover COVID-19 vaccines without cost-sharing, but that requirement will end after the federally declared public health emergency ends. 
However, a similar requirement does not apply to other Medicaid adults, and some states do not provide comprehensive vaccine coverage to these adults. States must cover vaccines without cost-sharing for children and for the Affordable Care Act Medicaid expansion population. In Medicaid, however, some enrollees might be left without vaccine coverage. In private plans and Medicare, that would continue even if the federal government stopped buying vaccines because both types of insurance coverage must cover vaccines at no cost (assuming lawmakers adopted the changes to facilitate Medicare coverage of vaccines available under EUAs that were described above). Require Medicaid coverage of COVID-19 vaccines without cost-sharing: Under the status quo, all insured patients can access COVID-19 vaccines at no cost.To avoid this, Congress could specify that COVID-19 products available under EUAs be treated as having full FDA approval for the purposes of the Medicare and Medicaid programs. At that point, there is a risk that some states, facing the prospect of paying full price for these products, would pare back their coverage beyond what would be permitted for drugs with full FDA approval. At present, other provisions of law require state Medicaid programs to cover all COVID-19 therapeutics, including those available solely under EUAs however, that requirement will end after the federally declared public health emergency ends. Importantly, these rules do not apply to vaccines, so lacking full FDA approval does not jeopardize a vaccine’s coverage in Medicaid, although other features of the Medicaid landscape may prevent coverage, an issue we return to below. In Medicaid, drugs available under EUAs fall outside Medicaid’s ordinary rules governing drug coverage, which, roughly speaking, require state Medicaid programs to cover all FDA approved drugs and require manufacturers to pay rebates under a statutory formula. Additionally, while Medicare Part B is currently covering COVID-19 vaccines available under EUAs (including the cost of administering the vaccine, all without patient cost-sharing), it is unclear whether that can continue after the federally declared public health emergency ends. Medicare Part D plans are generally not allowed to cover drugs that lack full FDA approval. However, both Medicare and Medicaid face legal obstacles to covering items available under EUAs. Furthermore, when new COVID-19 vaccines or therapeutics are developed in the future, it is likely that they will first become available under EUAs. Monoclonal antibody therapies, Pfizer’s Paxlovid, and Merck’s molnupiravir are also available only under EUAs. 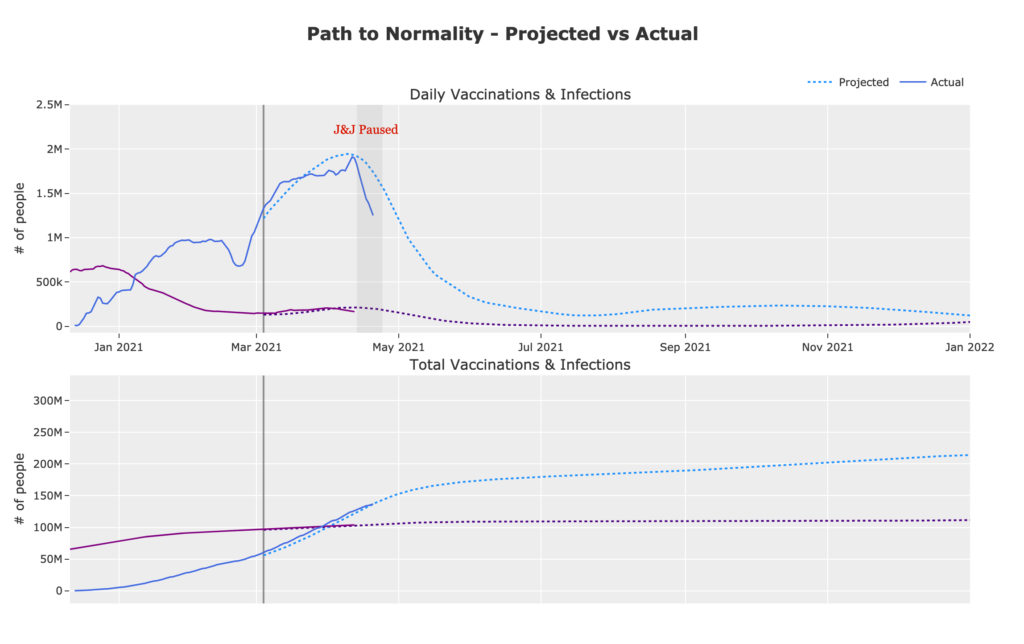
While the base two-dose series of the Pfizer and Moderna vaccines for adults now have full FDA approval, booster doses and vaccines for children are only available under EUAs. 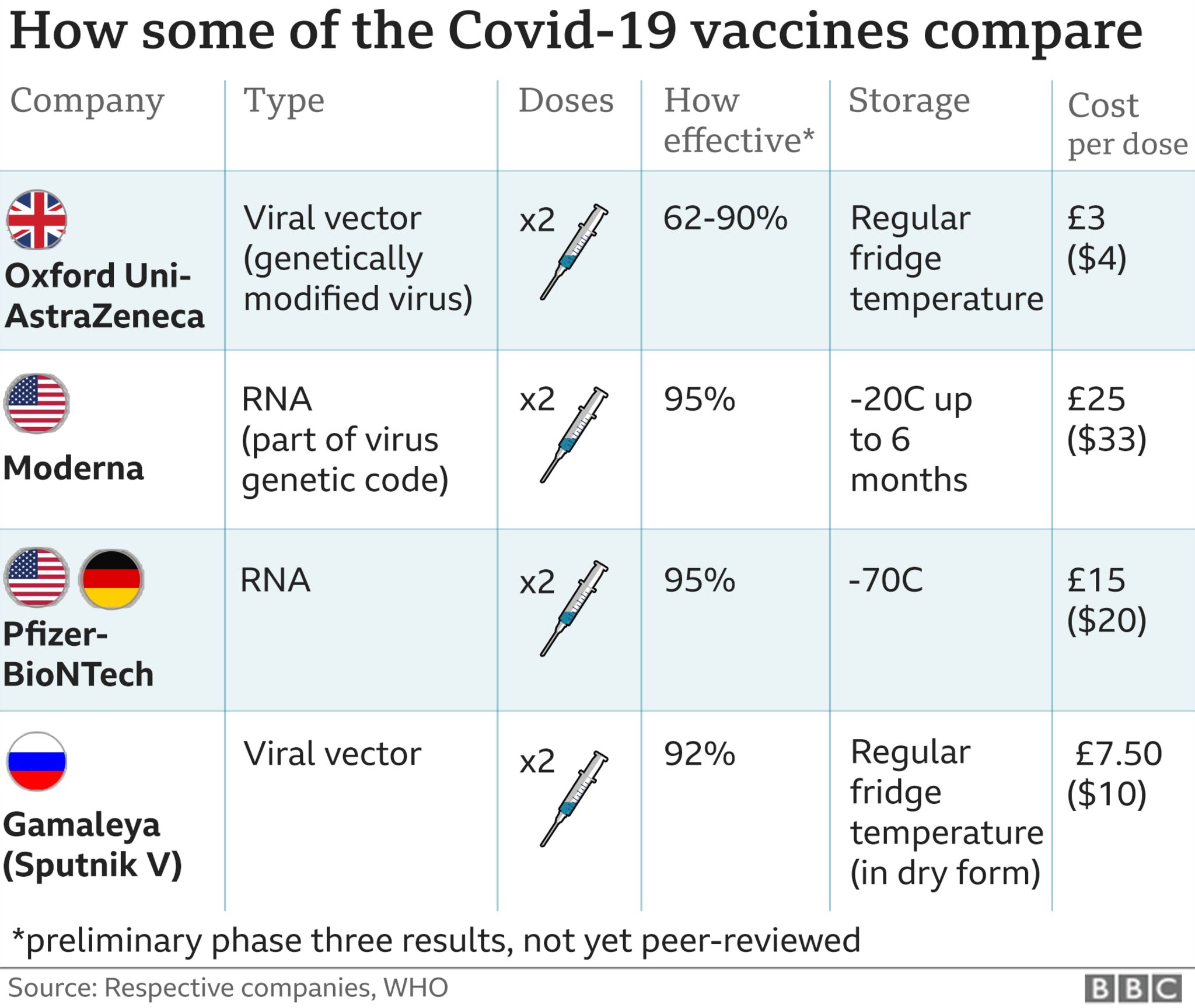
Facilitate Medicare and Medicaid coverage of items available under emergency use authorizations (EUAs): The FDA has made many COVID-19 vaccines and therapeutics available via EUAs rather than its regular approval process.This means that developing and manufacturing new vaccines and therapeutics-and ensuring continued easy access to existing ones-remains an urgent need. 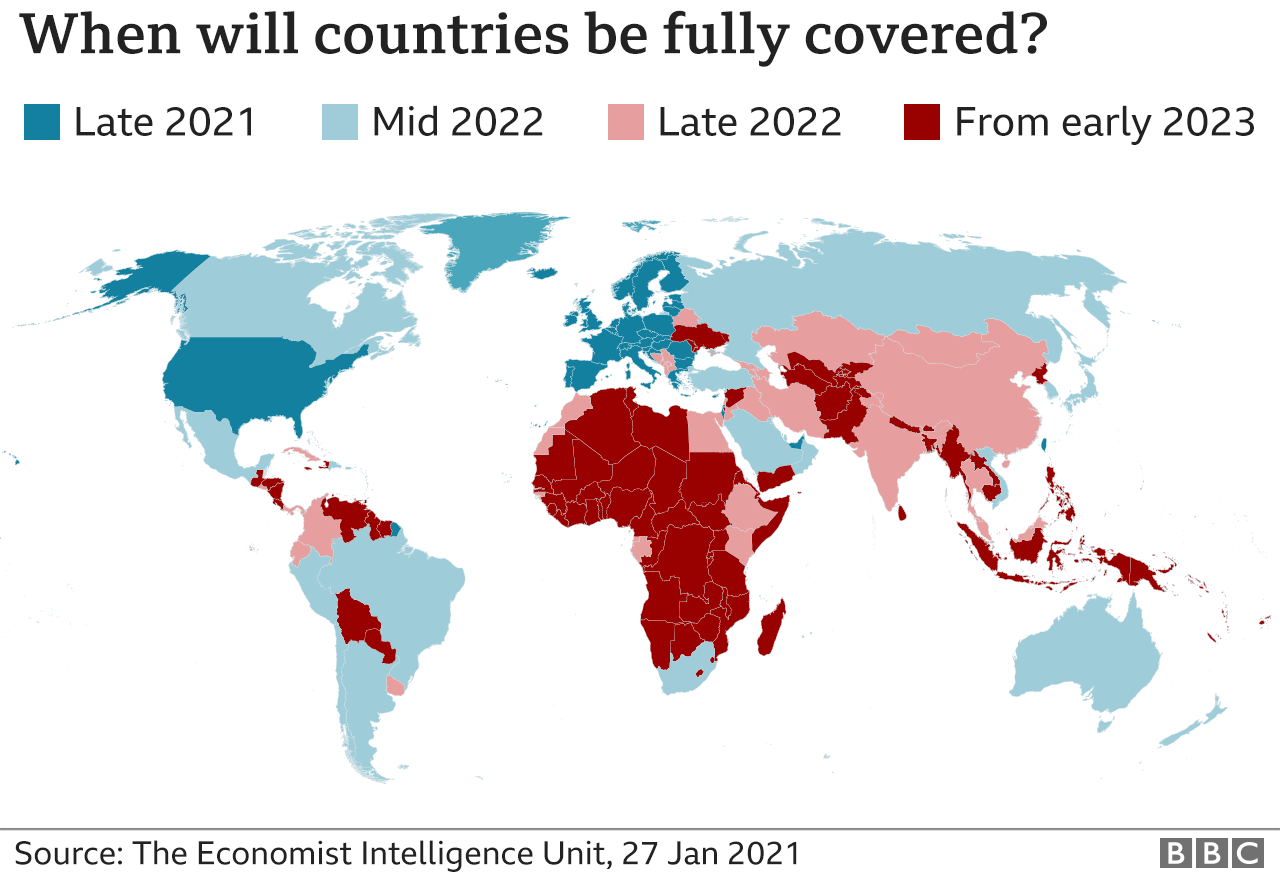
Moreover, the emergence of the Omicron variant showed that changes in the virus pose a serious threat to the effectiveness of existing vaccines and therapeutics: existing vaccines are less effective against Omicron, and some monoclonal antibody therapies are entirely ineffective. Existing vaccines, while highly effective, wane in effectiveness over time, so periodic booster doses will likely be needed for the foreseeable future indeed, older US adults are now being encouraged to receive a fourth dose. It is now clear that will not be the case. There may once have been hopes that a one-time vaccine rollout would produce durable herd immunity against COVID-19 in the United States, causing COVID-19 to fade into the background or disappear completely. A second lesson is that pandemic is dynamic, and what works today will not necessarily work tomorrow. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
